Research
Ken Field's Research Lab at Bucknell University
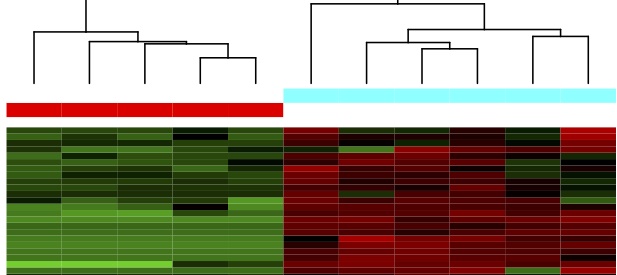
Immune responses in bats
Zoonotic diseases in bats
Fruit bats in Africa are reservoirs for many viral diseases that can spill over to humans including Ebola, Marburg, Nipah, and Hendra viruses. Students in the Field and Reeder labs are using next-generation RNA sequencing (RNA-Seq) to understand immune physiology and other metabolic processes within bats that contribute to disease spread. With support from the National Institutes of Health, we determine how differences in gene expression influence immune competence as a function of time of year, sex, age, and reproductive status. We will use this information to better understand how bats serve as disease vectors, to mitigate zoonotic disease outbreaks, and to prevent potential spillover events by predicting conditions that favor disease transmission.
 We are currently working in close collaboration with colleagues at Muni University in Arua, Uganda to study how different species of bats respond to viruses like Ebola. This work will help us understand why bats may serve as reservoir species and why they don’t appear to get sick from the same viruses that are deadly in humans. With the support of a $3m grant from the National Institutes of Health, we have established a research site at Muni University that allows us to study the immune responses of bats under field-like conditions.
We are currently working in close collaboration with colleagues at Muni University in Arua, Uganda to study how different species of bats respond to viruses like Ebola. This work will help us understand why bats may serve as reservoir species and why they don’t appear to get sick from the same viruses that are deadly in humans. With the support of a $3m grant from the National Institutes of Health, we have established a research site at Muni University that allows us to study the immune responses of bats under field-like conditions.
We are also interested in how bats respond to coronavirus infections and have received NSF support to determine if North American and African bat samples, collected for other studies, contain alpha- and beta- coronaviruses. By comparing the responses of bats, a natural host, to humans, a novel host, we hope to find differences that might explain why some COVID-19 infections are more pathological than others.
North American bats affected by white-nose syndrome
White-nose syndrome (WNS) has had catastrophic effects on bat populations in North America, with millions of bats from several species dying. Students in the Field and Reeder labs are studying how the cold-loving fungus that causes WNS, Pseudogymnoascus destructans (Pd), affects bats while they hibernate. By learning which specific types of immune responses Myotis lucifugus (little brown bats) can mount and the consequences of these immune responses, we hope to better understand bat immune physiology and the role of immune responses in WNS. With support from the US Fish and Wildlife Service, we hope that these studies will lead to the development of treatment strategies that can mitigate this potential extinction crisis. We are using a combination of approaches to determine how different bat species respond to Pd infection. Students in my lab are using whole-transcriptome approaches with next-generation sequencing (RNA-Seq). We also use quantitative PCR to measure the expression of specific genes and functional assays of memory immune responses. Because some species of bats are resistant to WNS (particularly European bats), we hope to identify protective responses that could then be augmented in susceptible species to promote WNS survival. We are also studying remnant bat populations in Pennsylvania to determine how some small groups of little brown bats have adapted to living with WNS.
 Hibernation and immune responses
Hibernation and immune responses
Bats are unique among mammals due to the metabolic demands of flight. In North America and Europe they are highly seasonal, using migration and/or hibernation to survive temperate winters. Bats are ecologically important animals because of their role as nocturnal predators of insects. For these reasons, I am interested in understanding the immune responses of bats and how their immune physiology compares to other hibernating and non-hibernating mammals. Together with the Reeder lab, my students are studying how hibernation affects immune function and gene expression in bats, mice, and thirteen-lined ground squirrels.
Undergraduate Research Projects
Bucknell students interested in conducting research with Prof. Field should contact him by e-mail (ken.field@bucknell.edu ) to arrange an appointment. Opportunities are available for summer research or for Biol 399 research. Most research projects span at least two semesters and the summer so interested students should become involved in their sophomore or junior year. Students are expected to complete Advanced Data Analysis in Biology (Biol 364) while enrolled in research.